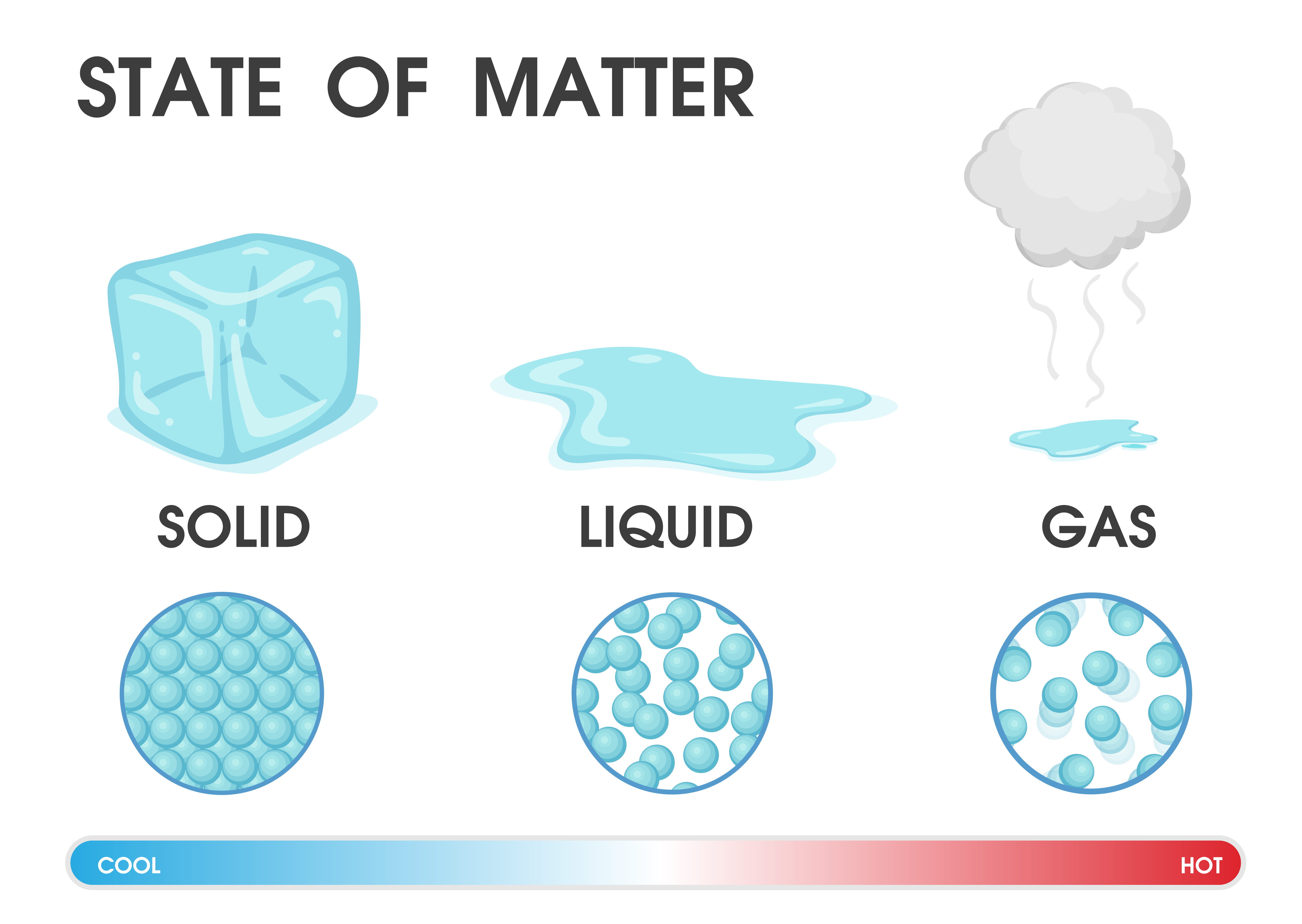
In 1811, Italian scientist Amedeo Avogadro proposed the idea that equal volumes of gas at the same temperature and pressure will have an equal number of particles, regardless of their chemical nature and physical properties. (Image credit: Luisa Vallon Fumi via Getty Images) Avogadro's number Joseph Louis Gay-Lussac collects air samples at different heights with Jean-Baptiste Biot in 1804. The law also explains why a turkey thermometer pops out when the turkey is done: The volume of air trapped under the plunger increases as the temperature inside the turkey climbs. Increasing temperature without increasing the volume available to accommodate the expanding gas means that pressure builds up inside the container and may cause it to explode. This law explains why it is an important safety rule that you should never heat a closed container. In order to keep the pressure constant, the volume of the container must be increased when a gas is heated. Heating a gas increases the kinetic energy of the particles, causing the gas to expand. This law states that the volume and temperature of a gas have a direct relationship: As temperature increases, volume increases when pressure is held constant. Most texts refer to this as Charles' law, but a few call it Gay-Lussac's law, or even the Charles Gay-Lussac law. In 1802, Joseph Louis Gay-Lussac, a French chemist and physicist referenced data gathered by his countryman, Jacque Charles, in a paper describing the direct relationship between the temperature and volume of a gas kept at a constant pressure. As long as the temperature stays the same, the pressure increases. As more air goes in, the gas molecules get packed together, reducing their volume. A good example of this is when you fill a tire with air. Increasing the amount of space available will allow the gas particles to spread farther apart, but this reduces the number of particles available to collide with the container, so pressure decreases.ĭecreasing the volume of the container forces the particles to collide more often, so the pressure is increased. Boyle's law states that if the temperature is held constant, volume and pressure have an inverse relationship that is, as volume increases, pressure decreases, according to the University of California, Davis' ChemWiki. Named after Robert Boyle, who first stated it in 1662. (Image credit: GeorgiosArt via Getty Images) 3.6: Changes in Matter - Physical and Chemical Changes Change is happening all around us all of the time.Chemist Robert Boyle stated that if the temperature is held constant, volume and pressure have an inverse relationship that is, as volume increases, pressure decreases.A chemical property describes the ability of a substance to undergo a specific chemical change. Physical properties include color, density, hardness, melting points, and boiling points. 3.5: Differences in Matter- Physical and Chemical Properties A physical property is a characteristic of a substance that can be observed or measured without changing the identity of the substance.A material composed of two or more substances is a mixture. A pure substance is a form of matter that has a consistent composition and properties that are constant throughout the sample. Matter can be classified into two broad categories: pure substances and mixtures. 3.4: Classifying Matter According to Its Composition One useful way of organizing our understanding of matter is to think of a hierarchy that extends down from the most general and complex, to the simplest and most fundamental.Liquids have a definite volume, but take the shape of the container. 3.3: Classifying Matter According to Its State-Solid, Liquid, and Gas Three states of matter exist-solid, liquid, and gas.Air (and all other gases) are invisible to the eye, have very small masses compared to equal amounts of solids and liquids, and are quite easy to compress (change volume). You might be able to imagine, however, the difficulty for people several hundred years ago to demonstrate that air has mass and volume. For most common objects that we deal with every day, it is fairly simple to demonstrate that they have mass and take up space. 3.2: What is Matter? Matter is anything that has mass and volume (takes up space).While this simple definition is easily applied, the way people view matter is often broken down into two characteristic length scales: the macroscopic and the microscopic. 
Matter includes atoms and anything made of atoms, but not other energy phenomena or waves such as light or sound. 3.1: In Your Room Matter is any substance that has mass and takes up space.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
